|
The cookie is used to store the user consent for the cookies in the category "Analytics". This cookie is set by GDPR Cookie Consent plugin. These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The particle model describes the energy, arrangement and movement of particles in solids, liquids and gases. This results in low numbers of particles in a given space. For example, liquid particles have fluidity, which means it is possible to make a liquid flow.Įxplanation: Particles have high energy and virtually no attraction between them therefore they are free to move apart. Solid particles vary considerably from liquids. How are liquid particles different from solid particles?Īlthough gases, liquids and solids feature atoms, molecules and sometimes ions as their key components, they have significant microscopic differences. The particles are not held in a fixed arrangement as in solids, so are able to slide past each other. Consequently, the particles are tightly packed. There is strong attraction between particles, but not as strong as in solids. How are particles held in relation to each other? A solid’s volume and shape are fixed, which means the particles are rigid and stay in place. While they do vibrate slightly, they do not move from place to place. 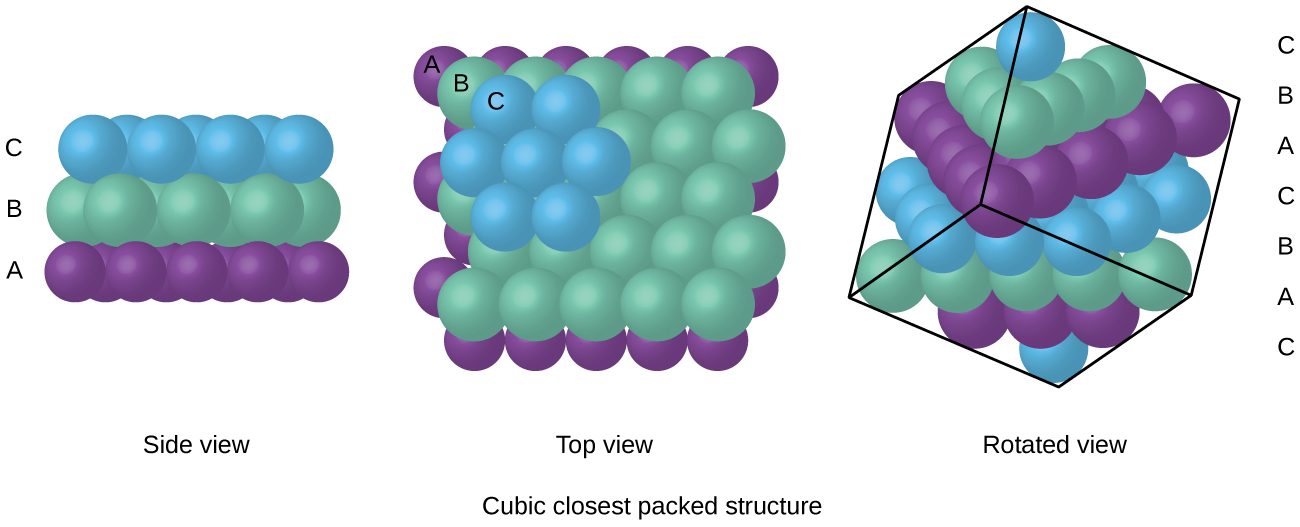 
Particles in a solid are usually packed close together, with a regular arrangement. The particles can move in their fixed positions but cannot slide past one another. Solid: Explain that in a solid, the particles are very attracted to each other so they are close together. Why are particles in a solid close together? solid are tightly packed, usually in a regular pattern. liquid are close together with no regular arrangement. Particles in a: gas are well separated with no regular arrangement. Which of the following is a correct particles arrangement of solid? The particles of such solids lack an ordered internal structure and are randomly arranged (Figure 1). When most liquids are cooled, they eventually freeze and form crystalline solids, solids in which the atoms, ions, or molecules are arranged in a definite repeating pattern. The gas particles have big distances between them.Īre particles in a solid randomly arranged? The particles in a liquid usually are still touching but there are some spaces between them. The particles in the solid are touching with very little space between them. The hotter a solid gets, the faster its particles vibrate.Īre the particles in a solid are spaced far apart? This gives solids a fixed shape and means that they cannot flow like liquids. The particles in solids move only by vibrating about a fixed position. 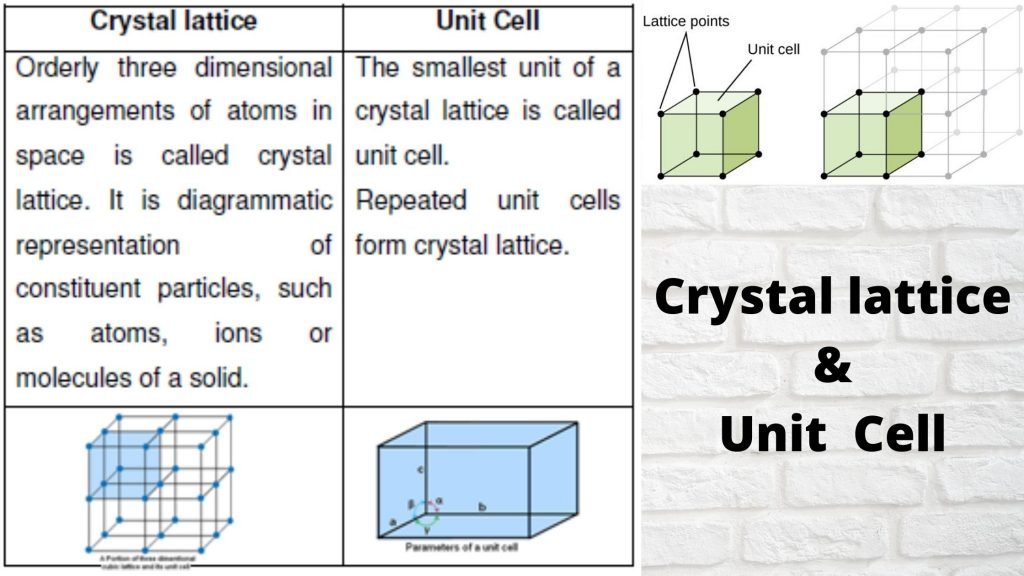
The particles in solids are arranged in a regular way. How are the particles arranged in a solid?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
